Unveiling the Neon Atom Model: A Closer Look
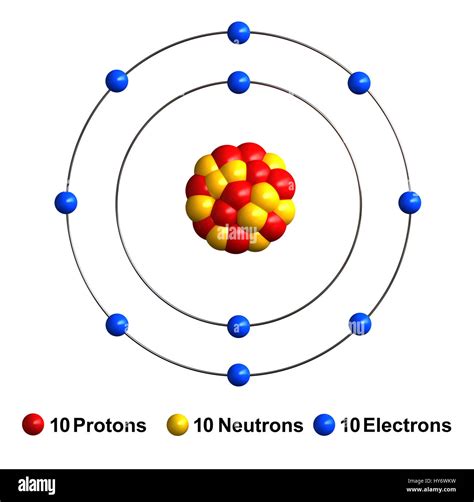
Unveiling the Neon Atom Model: A Closer Look
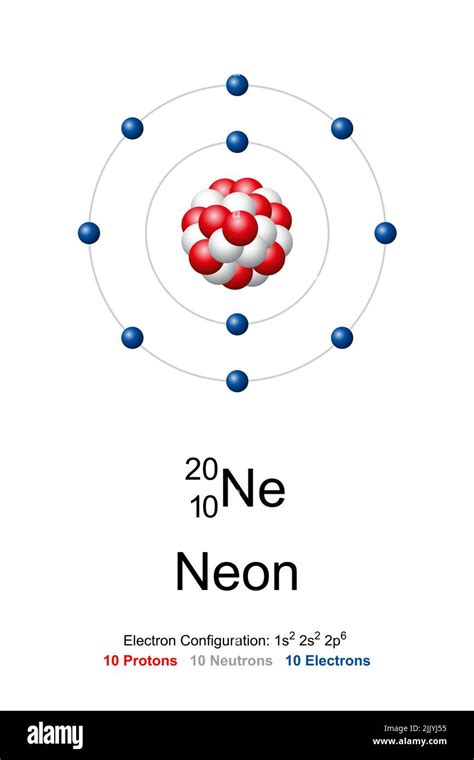
The neon atom model is a fascinating concept that has garnered significant attention in the realm of physics and chemistry. In this article, we will delve into the intricacies of the neon atom model, exploring its structure, properties, and applications.
Introduction to the Neon Atom Model

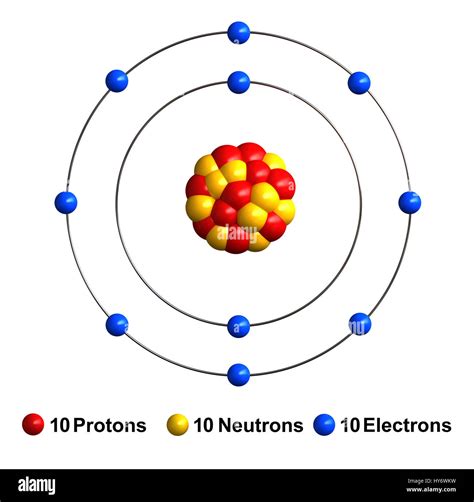
The neon atom model is a theoretical representation of the neon atom, which is a noble gas with the atomic number 10. The model is based on the Rutherford-Bohr model, which was developed by Ernest Rutherford and Niels Bohr in the early 20th century. The neon atom model consists of a nucleus surrounded by electrons, which are arranged in specific energy levels or shells.
Structure of the Neon Atom Model
The neon atom model is composed of a nucleus, which contains 10 protons and 10 neutrons. The nucleus is surrounded by 10 electrons, which are arranged in two energy levels or shells. The first energy level, also known as the 1s orbital, contains two electrons, while the second energy level, also known as the 2s and 2p orbitals, contains eight electrons.
| Energy Level | Electron Configuration |
|---|---|
| 1s | 2 electrons |
| 2s | 2 electrons |
| 2p | 6 electrons |

Properties of the Neon Atom Model
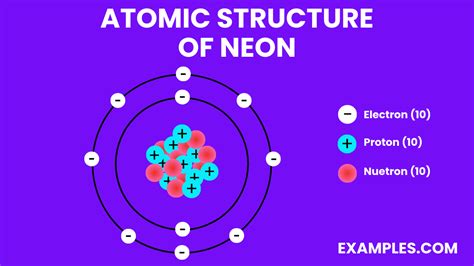
The neon atom model exhibits several interesting properties, including:
- Stability: The neon atom model is highly stable due to the full outer energy level, which makes it unreactive.
- Noble Gas Properties: The neon atom model exhibits typical noble gas properties, such as low reactivity and high ionization energy.
- Electron Configuration: The electron configuration of the neon atom model is 1s² 2s² 2p⁶, which is a stable configuration.
Applications of the Neon Atom Model

The neon atom model has several applications in various fields, including:
- Lighting: Neon is used in neon signs, which are widely used in advertising and decoration.
- Lasers: Neon is used in laser technology, particularly in the production of high-powered lasers.
- Plasma TVs: Neon is used in plasma TVs, which use individual cells filled with a gas, such as neon, to display images.
💡 Note: The neon atom model is a simplified representation of the neon atom and is not a realistic depiction of the actual atom.
In conclusion, the neon atom model is a fascinating concept that has contributed significantly to our understanding of the atomic structure and properties of noble gases. Its applications in various fields have revolutionized technology and continue to shape our daily lives.
What is the atomic number of neon?

+
The atomic number of neon is 10.
What is the electron configuration of the neon atom model?

+
The electron configuration of the neon atom model is 1s² 2s² 2p⁶.
What are some applications of the neon atom model?
+
The neon atom model has applications in lighting, lasers, and plasma TVs.



